PSUR repository updated
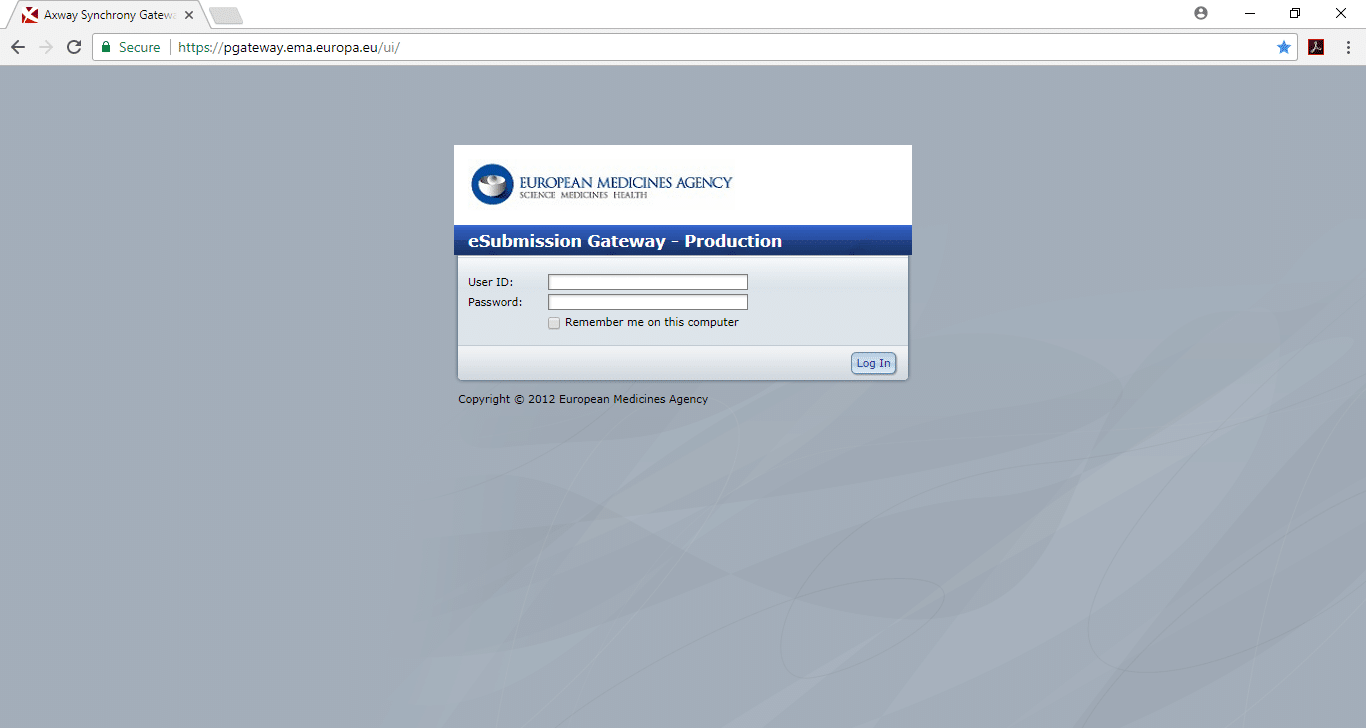
On the 26th of January 2015, the EMA introduced the pilot phase on the use of the XML delivery file for submissions of PSURs via the eSubmission Gateway/Web Client.
This introduction,
1) Simplified package file names for PSURs and
2) Introduced the requirement of an XML delivery file.
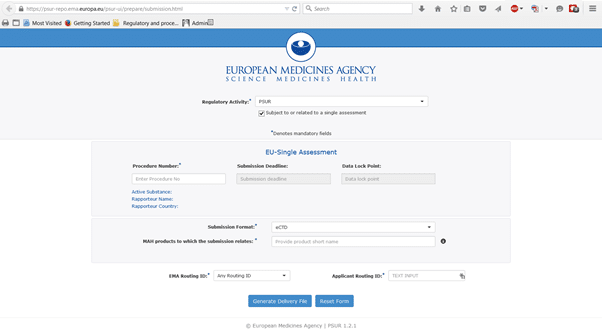
The XML delivery file is created in the PSUR Repository user interface
The PSUR Repository user interface is basically broken down into 4 sections:
- Section 1: Regulatory Activity
This is a simple drop down list and is currently only available for PSUR submissions and Supplementary information which is related to a PSUR submissions.
- Section 2: Details of assessment procedure
This section requires you to enter the Procedure Number as per the EURD list, all other information (Submission deadline, DLP, Active name, Rapporteur name and Rapporteur country) will prepopulate based on this Procedure Number. Carefully check if all information is correct, if prepopulated information is wrong contact the EMA.
- Section 3: Product Selection
- Select your Submission Format (eCTD or NeeS)
- Enter the product name(s) in the section products to which the submission relates for all your products to which the submission relates. This is a drop down menu based on the Art 57 database. Therefore you must first ensure that all your product information entered in the Art 57 database is up to date and correct.
- Select your products, enter your procedure number (where relevant) and the sequence number of your submission.
- Section 4 Routing information
- Enter EMA routing ID (e.g. ESUBPROD) and your Routing ID (e.g. ESUBPTESTPROD123)
- The XML delivery file will always be called xml, this should not be renamed/edited
- Save the XML delivery file inside the PSUR submission folder (zipped).
- Your PSUR folder should have a meaningful file name and examples of these can be found in Annex 3.
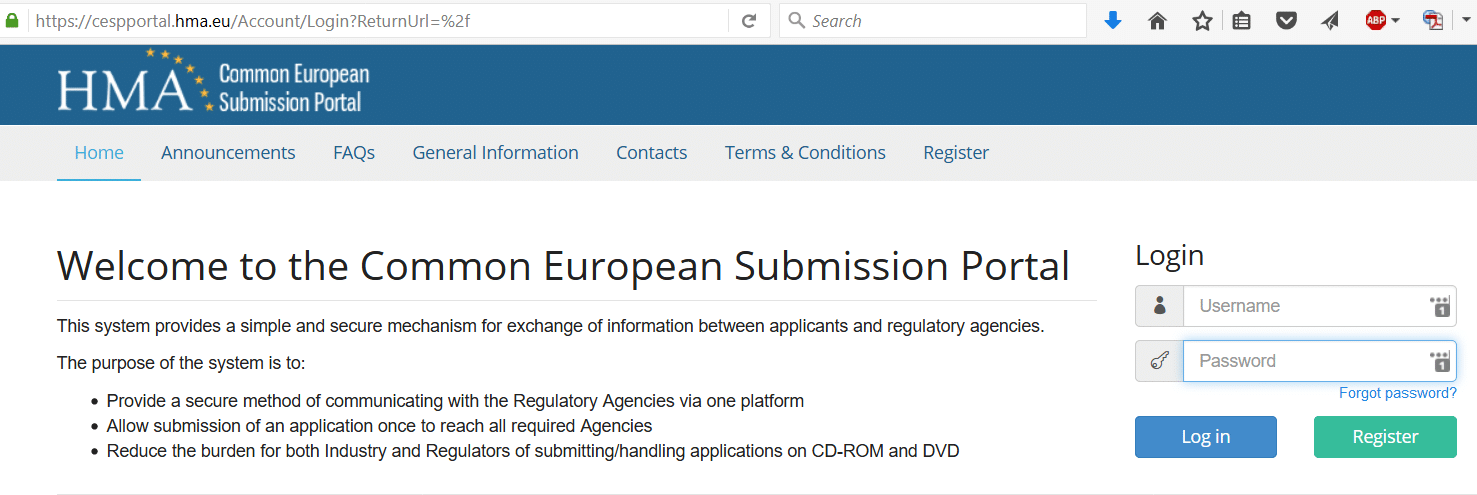
- Your Submission file can be send in the normal manner via the eSubmission Gateway/Web Client.
From September 2015, it will be mandatory to use the XML delivery file for all PSUR submission to the EMA via the eSubmission/Web Client.
Please contact us if you require any further information or help with this process.
Written by Fiona Downey.