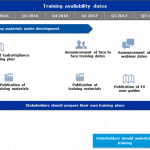
New ATC codes for 2017
The Anatomical Therapeutic Chemical (ATC) Classification is an internationally accepted classification system for medicines. This system is maintained by the World Health Organisation (WHO). The WHO assigns ATC codes to all drug substances based on their therapeutic indication. The assigned ATC code must be present in your product information.
What does this mean for you?
Marketing authorisation holders of medicinal products, authorised under the national/mutual recognition/decentralised and centralised procedures should be aware that some ATC codes have recently been changed.
Consequently, if changes have been made to your ATC code/text you must apply for an update of the Summary of Product Characteristics (namely section 5.1 Pharmacodynamic properties) via a Type IA A.6 variation.
Where can I find the ATC codes?
The updated ATC code for human and veterinary medicines can be found in the following links:
Human: https://www.whocc.no/atc_ddd_index/updates_included_in_the_atc_ddd_index/
Veterinary: https://www.whocc.no/atcvet/alterations_in_atcvet_codes/
We can help…
Ivowen are fully equipped to apply for such variations on your behalf. Please contact us for more information and for support of your dossier compilation or updates.
Written by Fiona Downey